Help me out here…
I was just reading the study Protection against SARS-CoV-2 after Covid-19 Vaccination and Previous Infection published in the NEJM March 31st 2022 by the SIREN study Group.
I quote the authors below:
Serum samples obtained from all the participants at baseline visits were collected centrally. These samples were tested at the U.K. Health Security Agency (formerly Public Health England) central testing laboratory at Porton Down with the use of the semiquantitative Elecsys Anti-SARS-CoV-2 nucleocapsid (N) protein assay and the fully quantitative Elecsys Anti-SARS-CoV-2 spike (S) protein assay (both manufactured by Roche Diagnostics).
I finally found that central lab ! This is where all the shenanigans are happening then…
Rare and Imported Pathogens Laboratory (RIPL)
UK Health Security Agency
Porton Down
Salisbury
Wiltshire SP4 0JG
United Kingdom
Looking at the users manual I read
In response to the coronavirus (COVID-19) pandemic, RIPL has developed capability for SARS CoV-2 PCR diagnostic testing and this is included in our current UKAS scope. Please contact the laboratory at portonswabtesting@phe.gov.uk if interaction with this service is required or desireable.
Testing utilises the COBAS 8800/6800 platforms and full details of acceptable sample types, turnaround times and reporting strategy will be provided on request.
So, on to my quest to check Roche Diagnostics
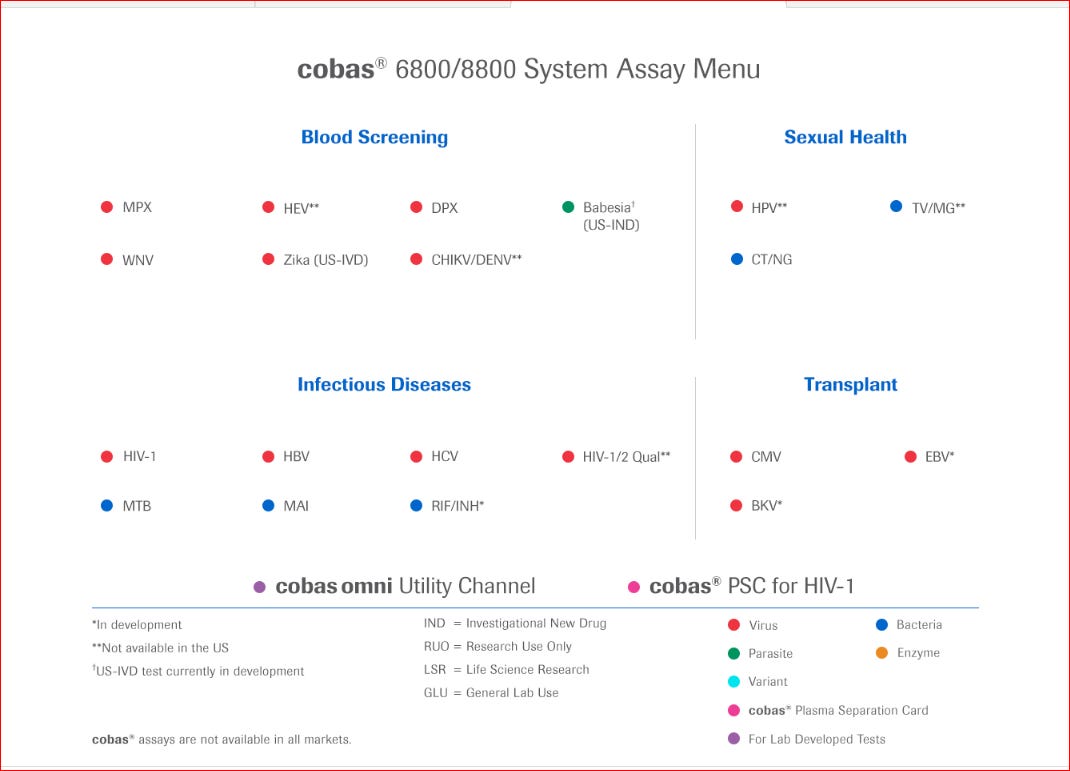
Roche Diagnostics market 4 assay systems; one of them being the COBAS 8800/6800 platform.
Let’s have a look at their platform and what it can do.
These are the assays performed in the platform.
Unless I have gone blind I cannot see the assay for SARS-CoV-2.
Can you?


Not a single virus has been isolated from an "infected" person.
If the "experts" don't have the "viruses," what the heck are they testing for?
It doesn't matter:
https://rayhorvaththesource.substack.com/p/who-is-right-and-who-is-wrong